True Value Dental Institute
True Value Dental Institute
Dry Socket Etiology, Diagnosis, and Clinical Treatment Techniques
Abstract
Dry socket, also termed fibrinolytic osteitis or alveolar osteitis, is a complication of tooth exodontia. A dry socket lesion is a post-extraction socket that exhibits exposed bone that is not covered by a blood clot or healing epithelium and exists inside or around the perimeter of the socket or alveolus for days after the extraction procedure. This article describes dry socket lesions; reviews the basic clinical techniques of treating different manifestations of dry socket lesions; and shows how microscope level loupe magnification of 6× to 8× or greater, combined with co-axial illumination or a dental operating microscope, facilitate more precise treatment of dry socket lesions. The author examines the scientific validity of the proposed causes of dry socket lesions (such as bacteria, inflammation, fibrinolysis, or traumatic extractions) and the scientific validity of different terminologies used to describe dry socket lesions. This article also presents an alternative model of what causes dry socket lesions, based on evidence from dental literature. Although the clinical techniques for treating dry socket lesions seem empirically correct, more evidence is required to determine the causes of dry socket lesions.
Keywords: Alveolar, Dry socket, Fibrinolysis, Osteitis.
I. Introduction and Definition of Dry Socket Lesions
The unscientific term “dry socket” refers to a post-extraction socket where some or all of the bone within the socket, or around the occlusal perimeter of the socket, is exposed in the days following the extraction, due to the bone not having been covered by an initial and persistent blood clot or not having been covered by a layer of vital, persistent, healing epithelium1,2. The patient may not be able to prevent food particles or the tongue from mechanically stimulating the exposed bone, which is acutely painful to touch, resulting in frequent acute pain. All parts of a dry socket lesion, except the exposed bone, can be gently touched with a periodontal probe or an irrigation needle tip without causing acute pain. Dry socket lesions occur in approximately 1% to 5% of all extractions and in up to 38% of mandibular third molar extractions1,2.
Food particles that collect inside the socket may dislodge a blood clot. Bacterial biofilm and food particles inside a socket may also hinder the reformation of a dislodged blood clot by obstructing contact of a reforming blood clot with the exposed bone. Food particles and bacterial biofilm may hinder contact of the healing epithelium with the exposed bone, which may prolong the healing time of the dry socket lesion. Food particles that collect inside a dry socket can also ferment due to bacteria. This fermentation may result in the formation of toxins or antigens that may irritate the exposed bone, produce an unpleasant taste or halitosis, and cause pain throughout the jaw. However, evidence suggests that bacteria is not the main cause of dry socket lesions1,2.
Microscope-level magnification of 6× to 8× or greater, combined with head-mounted or co-axial illumination, facilitates the observation of dry socket lesion anatomy such as exposed bone, either inside the socket or around the socket occlusal perimeter, areas of vital healing epithelium (which shows tensile strength when lightly probed), food particles or clumps of bacterial biofilm material within the socket, or inflamed gingival tissue, which may be sensitive to touch, but is not as sensitive as exposed bone.
This article presents a description and definition of the dry socket phenomenon, explores the proposed causes of dry sursocket lesions, and presents a comprehensive clinical approach to treating dry socket lesions, with an emphasis on how to achieve immediate coverage of exposed bone with such treatments. The author also presents a model of the causes of dry socket lesions based on current experimental knowledge. There is uncertainty in the dental literature about what causes dry socket lesions. Although some factors, such as smoking, oral contraceptive use, and presence of fibrinolytic activity in post-extraction sockets correlate with an increased incidence of dry socket, a definitive mechanism for explaining dry socket pathogenesis remains elusive1,2.
II. Treatment of Different Manifestations of Dry Socket Lesions
A dry socket lesion can present such that the bone inside the socket is exposed, but there is no exposed bone on the socket occlusal perimeter, and all of the exposed bone is below the projected location of the occlusal surface of the socket when the socket eventually heals.(Fig. 1) The socket bone can be completely exposed or can be covered by food debris or weakly clumped bacterial material. There may be some healing, which is exhibited by narrowing of the socket occlusal diameter by epithelial growth.

Fig. 1 A dry socket lesion where the socket perimeter is fully covered with healing epithelium, but a septum of exposed bone is visible inside the socket. The occlusal aspect of the septum bone is inferior to the projected plane of the occlusal aspect of the socket when the socket fully heals.
In this article, the basic treatment for dry sockets is to irrigate out food particles or bacterial material using chlorhexidine gluconate or saline and then fill the socket with a medicament1,3,4. (Fig. 2) The use of co-axial lighting and microscope-level magnification of 6× to 8× or greater facilitates the irrigation of a dry socket lesion and minimizes contact of the irrigation needle with exposed bone. Optimal visualization of the illuminated socket ensures that the irrigant reaches all the internal aspects of the socket and removes all microscopic debris. The dry socket medicament should cover the exposed bone for several days with a resorbable, but durable cover, which will protect the bone from painful mechanical stimulation, food impaction, and bacterial infiltration1. The dentist might suture the lesion to retain the medicament or blood clot and create a dense suture barrier over the socket opening if it is determined that chronic food impaction prevents systematic socket healing. The dentist may also anesthetize the patient and try to induce bleeding into the socket by aggressively curetting the socket or using a round bur or No. 330 bur with copious irrigation to avoid over-heating the bone to drill several 1.0 mm deep holes in the socket bone while avoiding arteries, nerves, thin socket walls, or other vulnerable anatomical features. When treating a dry socket lesion, the objective is to optimize the lesion such that the socket is optimally capable of forming an enduring layer of epithelium that covers the exposed bone inside the socket and around the socket occlusal perimeter.

Fig. 2 The dry socket lesion in Figure 1 after packing with an iodoform paste.
A dry socket lesion may show exposed bone located superior to the projected location of the occlusal surface of the socket after the socket heals. This bone may be a protruding septum of bone or may be located on the socket occlusal perimeter. This superiorly-located exposed bone would be the last aspect of the socket to be covered by epithelium, since the bone, protruding superiorly to the projected occlusal surface of the healed socket, would be exposed to food particles or mechanical trauma that may erode epithelium growing over that bone. This bone, if mechanically stimulated, would be a source of acute pain until the end of the healing period. A dentist may anesthetize the patient and use a football diamond bur with copious irrigation to trim this bone to approximately 1 mm inferior to the projected occlusal surface of the healed extraction socket. Such trimming can result in the bone becoming immediately coverable by a blood clot or medicament, thereby reducing the total number of days that this hyper-sensitive bone is exposed and helping to ensure that epithelium will systematically grow over the remaining exposed bone of the dry socket.
If the protruding bone is located on the socket occlusal perimeter, the dentist can reduce the bone to a level that is inferior to the occlusal aspect of the gingival tissue located just lateral to the protruding bone. If the gingiva on the socket occlusal perimeter is superior to all of the socket bone, a socket blood clot or dry socket medicament is more likely to cover the bone.
For some dry socket lesions, the dentist may observe and trim bone that protrudes buccally beyond the projected surface of the healed socket.(Fig. 3) Microscopes, combined with head-mounted co-axial illumination, facilitate the visualization of the interface between the protruding bone and the gingiva lateral to the protruding bone and result in selective drilling of the bone and not the gingiva.

Fig. 3 A dry socket lesion with separate buccal and occlusal areas of exposed bone.
A healing dry socket is a previous dry socket that is now completely covered with vital epithelium such that this epithelium covers all the socket bone and cannot be irrigated away.(Fig. 4) When a previous dry socket becomes completely epithelialized, this demonstrates that the socket has overcome mechanical stimulation or bacteria that were inhibiting the healing process. From this point, the socket will systematically progress toward complete healing, and the dry socket complication phase of the post-extraction healing process is over. As a result, the dentist no longer needs to debride the socket or apply medicament. The occlusal surface of a healing dry socket may be concave and collect food particles or plaque. If irrigation of bacterial material or food particles reveals a healthy layer of epithelium underneath, the bacteria or food particles are not preventing epithelialization of the socket. Any discomfort can be managed with non-narcotic analgesics; strong narcotic analgesics are not required. A chlorhexidine gluconate mouth rinse helps disinfect the socket while healing continues. A patient presenting with a healing dry socket may state that the socket had been uncomfortable in the past few days (when the socket was in the dry socket stage), but now feels better and simply wants the dentist to check that the socket is healing. A dentist can use microscopes and co-axial illumination to verify that a previous dry socket lesion is fully covered by epithelium by probing the epithelium to determine the presence of tensile strength, indicating vital tissue, and that there is no exposed bone that elicits acute pain to probing.
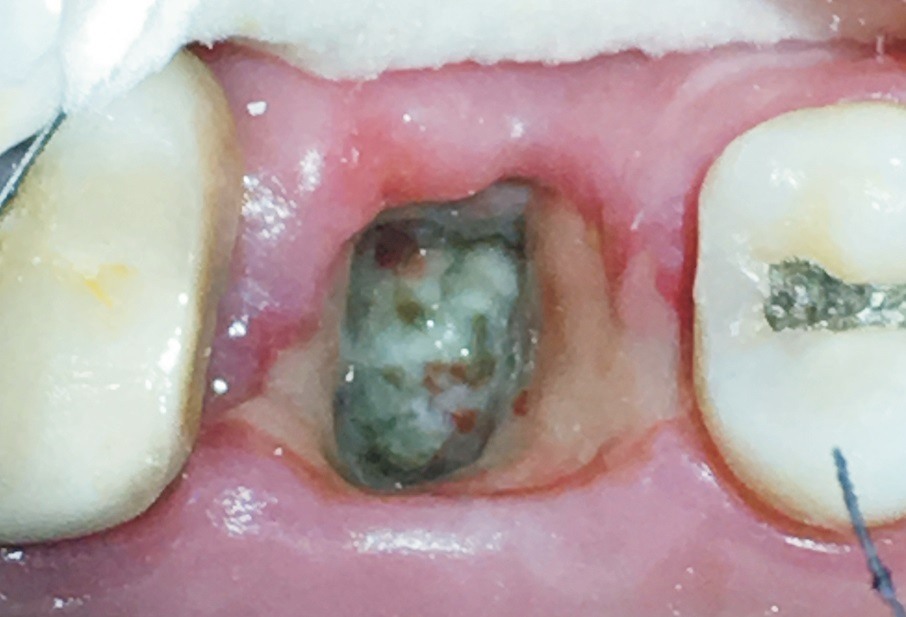
Fig. 4 Example of a previous dry socket lesion that is now fully covered with a layer of epithelium that does not wash away with irrigation.
III. Proposed Causes of Dry Socket Lesions
Comprehensive reviews of the proposed causes of dry socket lesions and of the factors that correlate with increased dry socket incidence can be found in the literature1,2,5,6,7,8,9. One hypothesis is that bacteria initiate dry socket lesions or prolong their duration1,2,5,6,7,8,9,10. However, there is little evidence that antibiotics given after an extraction reduce dry socket incidence11,12,13. An antiseptic Chlorhexidine gel, placed prophylactically in extraction sockets after the procedure, does not significantly reduce dry socket incidence14,15. However, one meta-analysis found that systemic antibiotics given before third molar surgery reduced dry socket incidence16. Overall, these findings suggest that reducing bacterial counts around extraction sockets may only result in an insignificant reduction in dry socket incidence.
IV. Proposed Model of Dry Socket Lesion Pathogenesis
A model of dry socket lesion pathogenesis can explain various facts about dry sockets including the findings that smoking2,17,18 and use of oral contraceptives2,18 increase the incidence of dry socket lesions. In addition, the model can also demonstrate that there can be a 24- to 96-hour delay after an extraction before dry socket lesions appear2,5; that traumatic extractions, where heavy luxation or forceps forces are required to extract teeth particles, increase the incidence of dry socket lesions19; that plasmin-induced fibrinolysis activity seems higher in dry socket lesions compared to non-dry-socket post-extraction sockets2,6,9; and that bacteria do not seem to initiate dry socket lesions11,12,13. Such a model should explain whether or not inflammation causes dry socket lesions.
Birn observed high concentrations of plasmin and increased fibrinolytic activity in the alveolar bone lining dry socket lesions6,9. Plasminogen, the precursor of plasmin, circulates in the blood and binds to clots at wound sites. Various tissue activators, including tissue-type and urokase-type plasminogen activators20,21, convert plasminogen to plasmin6,20,21,22. Plasmin is experimentally identified as an important molecule for inducing inflammation20,22,23,24 because it has been found to induce fibrinolysis to dissolve blood vessel clots, increase local capillary permeability, and attract inflammatory cells and its complements to wound sites.
Birn hypothesized that trauma during an extraction or the presence of a bacterial infection somehow facilitates the release of plasminogen tissue activators in the post-extraction socket, resulting in the plasmin induction of fibrinolysis that dislodges the blood clot that formed after the extraction and causing a dry socket lesion6,9. However, although Birn found a correlation between the presence of fibrinolytic activity in extraction sockets and dry socket lesion pathogenesis, fibrinolysis may not be the cause dry socket lesions. Since fibrinolysis also increases capillary blood flow to the extraction socket, it might actually reduce the probability of dry socket lesion formation25,26,27,28. Dry socket lesions routinely exhibit an eventual stoppage of blood flow to the socket. This idiopathic ischemia counteracts the effect of fibrinolysis and is presumably a cause of dry socket lesion initiation and pathogenesis.
As an alternative to Birn’s fibrinolytic theory, the author proposes a different model of dry socket lesion initiation and pathogenesis. In a high-stress extraction, that puts high compressive forces on alveolar bone surrounding the tooth, events are initiated that will cause, over a 24- to 96-hour period following the extraction, the necrosis of osteoblasts lining the intaglio surface of the socket. The necrosis of the osteoblasts may initiate fibrinolytic activity that lyses any blood clot that may have formed after the extraction, or the blood clot may dislodge because the necrotic osteoblasts lose the ability to metabolically integrate with the blood clot. Also, approximately at the time of osteoblast necrosis, the socket stops bleeding, even though the fibrinolytic activity should theoretically cause increased bleeding to the extraction socket to bring immune cells and complements to the socket to begin resorbing the necrotic osteoblasts. This idiopathic socket ischemia event may prevent an initial blood clot to reform through additional bleeding and may prevent the immune system from accessing the site through local capillaries to initiate an inflammatory response to resorb the necrotic bone cells. The necrotic bone cells are then exposed and uncovered for several days, resulting in the major symptom (or morbidity) of dry socket lesions, acute pain of the exposed socket to mechanical stimulation that lingers for several days until the bone becomes completely covered by healing epithelium.
During a traumatic extraction, heavy luxation or forceps forces transfer to the jawbone surrounding the roots and may crush bone on the intaglio surface of the extraction socket1,10,29. This can induce necrosis or apoptosis of osteoblasts within the extraction socket30,31,32. Studies have shown that mechanical stress (excess tensile or compression forces) on osteoblasts can activate cellular signaling pathways that lead to osteoblast apoptosis30,31,32,33. Also, the percentage of apoptotic osteoblasts increases over 24 hours after the initial compressive force application30 and increases in proportion to the compressive force30,33.
The necrosis of bone cells, occurring over a >24-hour delay period after an extraction, may result in the bone cells releasing urokinase plasminogen tissue activator, which is the main plasminogen activator released in dry socket lesions21. The urokinase plasminogen tissue activator then converts plasminogen to plasmin. The plasmin may directly result in the lysis of a blood clot that initially formed in the socket. However, a major function of plasmin is to initiate blood vessel perfusion to bring blood, immune system cells, and complements to the intaglio surface of the socket to begin resorbing the necrotic osteoblasts. In dry socket lesions, however, an idiopathic blood vessel ischemia event is eventually observed that prematurely blocks this capillary perfusion-mediated immune system activation process.
The cause of ischemia at a dry socket lesion site is unknown. Theoretically, the high forces of the extraction may crush and occlude blood vessels within the bone forming the intaglio surface of the socket (although there is no experimental evidence for or against compression-induced blood vessel occlusion existing in dry socket lesions). Some socket bone may be dense, with few blood vessels per unit of socket area, or a socket may be observed to only bleed from the apical aspect, making these sockets intrinsically incapable of significant bleeding. Smoking or oral contraceptive use may also reduce systemic blood circulation17,18. In addition, the pro-bleeding effect of plasminolysis may be counteracted chemically by pro-ischemia thrombin activity34 at the dry socket wound site.
Due to the lack of blood flow to the intaglio surface of the socket, the immune system cells and their complement factors cannot be brought to the intaglio surface of the socket to resorb the necrotic bone cells lining the socket. Instead, clinical observation seems to show that the socket heals by a mechanism where vital epithelium, initially present at the outer perimeter of the socket, grows gradually from the outer perimeter of the socket inferiorly into the socket down to the apex of the socket. As the vital epithelium gradually covers the surface area of the socket intaglio surface, the epithelium brings blood vessels, immune system cells, and their complements in direct contact with the necrotic bone cells of the socket to begin resorbing the necrotic bone cells. This process of epithelium growth may take several days; during this time, the uncovered bone is painful to the touch and is vulnerable to painful contact with bacterial biofilm or food impaction.
This model of dry socket pathogenesis and healing implies that inflammation does not fundamentally cause dry socket lesions and is not the cause of dry socket morbidity (Fig. 5) because ischemia will prevent an inflammatory event from occurring at the dry socket lesion site. Therefore, this model questions the use of terminology such as “alveolar osteitis,” or “fibrinolytic osteitis,” or any other term using the inflammation suffix “-itis” to describe dry socket lesions. Instead, the author suggests an alternative terminology for the dry socket phenomenon: “post-extraction peri-alveolar exposed-bone ostealgia syndrome.”

Fig. 5 Example of a maxillary posterior dry socket lesion surrounded by a viral outbreak. Although the outbreak may theoretically increase generalized inflammation around the dry socket, it is unknown if the outbreak increases pain or the duration of the dry socket or is only coincident with the lesion.
V. Evidence for the Model of Dry Socket Lesion Pathogenesis
There is evidence that reduced post-extraction socket blood flow facilitates dry socket lesion formation. Smoking17,18 and use of oral contraceptives18 both facilitate blood clotting throughout the body35 and may reduce blood circulation into the extraction socket. Both smoking and use of oral contraceptives correlate with an increased incidence of dry socket lesions2.
Traumatic extractions correlate with dry socket lesion incidence19. The incidence of dry socket lesions is lower for non-surgical extractions (that do not require tooth sectioning) compared to surgical extractions15,18,36,37. This may be due to a correlation between the need to section a tooth and the need for heavy luxation forces to remove a tooth or individual roots.
The highest rate of dry socket incidence among all teeth types occurs with the extraction of mandibular third molars. Mandibular third molars are often deeply embedded in dense bone and have the highest incidence of root dilacerations among teeth38,39,40. Mandibular third molars may have roots that are not radially co-axial with the imaginary radial axis on which the dentist places luxation forces to remove the root, especially if difficult access limits the number of possible ways of positioning luxation instruments. These factors may obligate a dentist to use heavy forceps or luxation forces, even after root sectioning, to extract mandibular third molars, and these heavy forces may transmit to the surrounding jawbone. Crawford41 first described dry socket lesions, using a case report where he extracted a mandibular third molar “with great difficulty,” and may not have sectioned the tooth, given the limited technologies in 1896.
The incidence of dry socket lesion formation is lower with maxillary third molar extractions compared to mandibular third molar extractions. Maxillary third molars often have conical roots embedded in cancellous bone bounded by thin buccal bone, requiring less force for removal. Extracting teeth that are in cancellous bone may result in multiple sharp points of cancellous bone severing multiple blood vessels, which may ensure bleeding into the post-extraction socket and blood clot formation.
VI. Scenarios That Result in High Stress Extractions
One example of a low stress extraction is if the dentist sections teeth before attempting extraction using heavy luxation and forceps forces. Also, infected teeth where the periodontal ligament has been resorbed by an underlying abscess can often be extracted with minimal stresses on surrounding alveolar bone, even if the roots feature ellipsoid cross sections. However, various situations can result in a tooth extraction where heavy stresses are placed on the surrounding jawbone:
- A dentist may extract a multi-rooted tooth using heavy luxation and forceps forces, moving the tooth back and forth to expand the socket to facilitate tooth extraction without sectioning the tooth roots that may be interlocked in bone.
- A dentist may initially attempt to extract a multi-rooted tooth using heavy luxation and forceps forces, but after placing heavy forces on the tooth, decides to section the tooth39. Sectioning the tooth results in less force needed to extract the tooth, but the heavy forces placed on the tooth prior to sectioning stressed the jawbone.
- A dentist sections a multi-rooted tooth prior to placing heavy luxation or forceps forces on the tooth. However, the sectioned individual roots still require heavy luxation forces to extract them. This often occurs when extracting endodontically treated roots that may be partially or fully ankylosed within the surrounding alveolar bone.
- Teeth with ellipsoid cross sections (particularly maxillary canines and two-rooted maxillary premolars) often cannot be extracted by rotating in a superior direction inside the socket using forceps, unless heavy forces are used. A root may be difficult to extract if it has an hour-glass cross-sectional shape due to mesial and distal concavities or if the root is ankylosed due to endodontic treatment. The dentist may be able to extract ellipsoid roots with minimal stress on the surrounding alveolar bone by sectioning the coronal 2/3 of the root mid-way between the buccal and lingual aspects of the root or by removing bone that has grown into the mesial and distal root concavities of the root to create circular cross sections of the sectioned teeth fragments.
VII. Conclusion
This article described different manifestations of dry socket lesions, summarized the treatment approaches for each different manifestation, reviewed the proposed causes of dry socket lesions, described and presented a model of dry socket lesion pathogenesis, and proposed a different terminology for the dry socket phenomenon. More evidence is needed to prove the scientific validity of techniques of dry socket lesion treatment, to validate the proposed model, and to determine which factors cause dry socket lesions.
Footnotes
Contributed by
Author’s Contributions: J.M. developed the article concept, took clinical photographs, performed background research and wrote the manuscript.
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
1. Bowe DC, Rogers S, Stassen LF. The management of dry socket/alveolar osteitis. J Ir Dent Assoc. 2011–2012;57:305–310. [PubMed] [Google Scholar]
2. Blum IR. Contemporary views on dry socket (alveolar osteitis): a clinical appraisal of standardization, aetiopathogenesis and management: a critical review. Int J Oral Maxillofac Surg. 2002;31:309–317. [PubMed] [Google Scholar]
3. Taberner-Vallverdú M, Nazir M, Sánchez-Garcés MÁ, Gay-Escoda C. Efficacy of different methods used for dry socket management: a systematic review. Med Oral Patol Oral Cir Bucal. 2015;20:e633–e639. [PMC free article] [PubMed] [Google Scholar]
4. Daly B, Sharif MO, Newton T, Jones K, Worthington HV. Local interventions for the management of alveolar osteitis (dry socket) Cochrane Database Syst Rev. 2012;12:CD06968 [PubMed] [Google Scholar]
5. Nitzan DW. On the genesis of “dry socket” J Oral Maxillofac Surg. 1983;41:706–710. [PubMed] [Google Scholar]
6. Birn H. Etiology and pathogenesis of fibrinolytic alveolitis (“dry socket”) Int J Oral Surg. 1973;2:211–263. [Google Scholar]
7. Birn H. Bacteria and fibrinolytic activity in “dry socket” Acta Odontol Scand. 1970;28:773–783. [PubMed] [Google Scholar]
8. Birn H. Fibrinolytic activity of normal alveolar bone. Acta Odontol Scand. 1971;29:141–153. [PubMed] [Google Scholar]
9. Birn H. Fibrinolytic activity in “dry socket” Acta Odontol Scand. 1970;28:37–58. [PubMed] [Google Scholar]
10. Colby RC. The general practitioner’s perspective of the etiology, prevention, and treatment of dry socket. Gen Dent. 1997;45:461–467. quiz 471-2. [PubMed] [Google Scholar]
11. Olurotimi AO, Gbotolorun OM, Ibikunle AA, Emeka CI, Arotiba GT, Akinwande JA. A comparative clinical evaluation of the effect of preoperative and postoperative antimicrobial therapy on postoperative sequelae after impacted Mandibular third molar extraction. J Oral Maxillofac Res. 2014;5:e2 [PMC free article] [PubMed] [Google Scholar]
12. Lee JY, Do HS, Lim JH, Jang HS, Rim JS, Kwon JJ, et al. Correlation of antibiotic prophylaxis and difficulty of extraction with postoperative inflammatory complications in the lower third molar surgery. Br J Oral Maxillofac Surg. 2014;52:54–57. [PubMed] [Google Scholar]
13. Reekie D, Downes P, Devlin CV, Nixon GM, Devlin H. The prevention of ‘dry socket’ with topical metronidazole in general dental practice. Br Dent J. 2006;200:210–213. [PubMed] [Google Scholar]
14. Freudenthal N, Sternudd M, Jansson L, Wannfors K. A double-blind randomized study evaluating the effect of intra-alveolar chlorhexidine gel on alveolar osteitis after removal of mandibular third molars. J Oral Maxillofac Surg. 2015;73:600–605. [PubMed] [Google Scholar]
15. Abu-Mostafa NA, Alqahtani A, Abu-Hasna M, Alhokail A, Aladsani A. A randomized clinical trial compared the effect of intraalveolar 0.2% Chlorohexidine bio-adhesive gel versus 0.12% Chlorohexidine rinse in reducing alveolar osteitis following molar teeth extractions. Med Oral Patol Oral Cir Bucal. 2015;20:e82–e87. [PMC free article] [PubMed] [Google Scholar]
16. Ren YF, Malmstrom HS. Effectiveness of antibiotic prophylaxis in third molar surgery: a meta-analysis of randomized controlled clinical trials. J Oral Maxillofac Surg. 2007;65:1909–1921. [PubMed] [Google Scholar]
17. Meechan JG, Macgregor ID, Rogers SN, Hobson RS, Bate JP, Dennison M. The effect of smoking on immediate post-extraction socket filling with blood and on the incidence of painful socket. Br J Oral Maxillofac Surg. 1988;26:402–409. [PubMed] [Google Scholar]
18. Abu Younis MH, Abu Hantash RO. Dry socket: frequency, clinical picture, and risk factors in a palestinian dental teaching center. Open Dent J. 2011;5:7–12. [PMC free article] [PubMed] [Google Scholar]
19. Haraji A, Rakhshan V. Single-dose intra-alveolar chlorhexidine gel application, easier surgeries, and younger ages are associated with reduced dry socket risk. J Oral Maxillofac Surg. 2014;72:259–265. [PubMed] [Google Scholar]
20. Medcalf RL. Fibrinolysis, inflammation, and regulation of the plasminogen activating system. J Thromb Haemost. 2007;(5 Suppl 1):132–142. [PubMed] [Google Scholar]
21. Serratì S, Margheri F, Bruschi S, D’Alessio S, Pucci M, Fibbi G, et al. Plasminogen activators and inhibitor type-1 in alveolar osteitis. Eur J Oral Sci. 2006;114:500–503. [PubMed] [Google Scholar]
22. Berri F, Rimmelzwaan GF, Hanss M, Albina E, Foucault-Grunenwald ML, Lê VB, et al. Plasminogen controls inflammation and pathogenesis of influenza virus infections via fibrinolysis. PLoS Pathog. 2013;9:e1003229. [PMC free article] [PubMed] [Google Scholar]
23. Syrovets T, Lunov O, Simmet T. Plasmin as a proinflammatory cell activator. J Leukoc Biol. 2012;92:509–519. [PubMed] [Google Scholar]
24. Li Q, Laumonnier Y, Syrovets T, Simmet T. Plasmin triggers cytokine induction in human monocyte-derived macrophages. Arterioscler Thromb Vasc Biol. 2007;27:1383–1389. [PubMed] [Google Scholar]
25. Moore EE, Moore HB, Gonzalez E, Chapman MP, Hansen KC, Sauaia A, et al. Postinjury fibrinolysis shutdown: rationale for selective tranexamic acid. J Trauma Acute Care Surg. 2015;78(6 Suppl 1):S65–S69. [PMC free article] [PubMed] [Google Scholar]
26. Orsi FA, Angerami RN, Mazetto BM, Quaino SK, Santiago-Bassora F, Castro V, et al. Reduced thrombin formation and excessive fibrinolysis are associated with bleeding complications in patients with dengue fever: a case-control study comparing dengue fever patients with and without bleeding manifestations. BMC Infect Dis. 2013;13:350. [PMC free article] [PubMed] [Google Scholar]
27. van Herrewegen F, Meijers JC, Peters M, van Ommen CH. Clinical practice: the bleeding child. Part II: disorders of secondary hemostasis and fibrinolysis. Eur J Pediatr. 2012;171:207–214. [PMC free article] [PubMed] [Google Scholar]
28. Chapin JC, Hajjar KA. Fibrinolysis and the control of blood coagulation. Blood Rev. 2015;29:17–24. [PMC free article] [PubMed] [Google Scholar]
29. Halabí D, Escobar J, Muñoz C, Uribe S. Logistic regression analysis of risk factors for the development of alveolar osteitis. J Oral Maxillofac Surg. 2012;70:1040–1044. [PubMed] [Google Scholar]
30. Goga Y, Chiba M, Shimizu Y, Mitani H. Compressive force induces osteoblast apoptosis via caspase-8. J Dent Res. 2006;85:240–244. [PubMed] [Google Scholar]
31. Matsui H, Fukuno N, Kanda Y, Kantoh Y, Chida T, Nagaura Y, et al. The expression of Fn14 via mechanical stress-activated JNK contributes to apoptosis induction in osteoblasts. J Biol Chem. 2014;289:6438–6450. [PMC free article] [PubMed] [Google Scholar]
32. Nettelhoff L, Grimm S, Jacobs C, Walter C, Pabst AM, Goldschmitt J, et al. Influence of mechanical compression on human periodontal ligament fibroblasts and osteoblasts. Clin Oral Investig. 2016;20:621–629. [PubMed] [Google Scholar]
33. Hu K, Wang C, Zhang X. High pressure may inhibit periprosthetic osteogenesis. J Bone Miner Metab. 2010;28:289–298. [PubMed] [Google Scholar]
34. Delvaeye M, Conway EM. Coagulation and innate immune responses: can we view them separately. Blood. 2009;114:2367–2374. [PubMed] [Google Scholar]
35. Bonnar J. Coagulation effects of oral contraception. Am J Obstet Gynecol. 1987;157:1042–1048. [PubMed] [Google Scholar]
36. Parthasarathi K, Smith A, Chandu A. Factors affecting incidence of dry socket: a prospective community-based study. J Oral Maxillofac Surg. 2011;69:1880–1884. [PubMed] [Google Scholar]
37. Torres-Lagares D, Serrera-Figallo MA, Romero-Ruíz MM, Infante-Cossío P, García-Calderón M, Gutiérrez-Pérez JL. Update on dry socket: a review of the literature. Med Oral Patol Oral Cir Bucal. 2005;10:81–85. [PubMed] [Google Scholar]
38. Hamasha AA, Al-Khateeb T, Darwazeh A. Prevalence of dilaceration in Jordanian adults. Int Endod J. 2002;35:910–912. [PubMed] [Google Scholar]
39. Malcić A, Jukić S, Brzović V, Miletić I, Pelivan I, Anić I. Prevalence of root dilaceration in adult dental patients in Croatia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102:104–109. [PubMed] [Google Scholar]
40. Colak H, Bayraktar Y, Hamidi MM, Tan E, Colak T. Prevalence of root dilacerations in Central Anatolian Turkish dental patients. West Indian Med J. 2012;61:635–639. [PubMed] [Google Scholar]
41. Crawford JY. Dry socket. Dental Cosmos. 1896;38:929–931. [Google Scholar]

 Click here for Dental Services
Click here for Dental Services